A paper describing the death of a young woman trying to lose weight by consuming Herbalife® products has been withdrawn after the company threatened to sue the journal.

Herbalife and criticism
Herbalife Nutrition develops and sells plant-based dietary supplements for weight loss. According to Wikipedia, 58 percent of their shakes, protein bars, teas, and other products are made in company-owned manufacturing facilities in the US and China, with the remainder manufactured by third-party contractors.
In 2016 the FTC filed a complaint that the company was being run as a “pyramid scheme.” The complaint was settled for $200 million.
In addition the company has been criticized for the accuracy of their claims about health benefits, and also for the presence of lead and other toxic ingredients in several of their products. The Herbalife Wikipedia page lists hospitals all over the world that have reported liver damage in patients using Herbalife products.
The paper
Philips CA et al. REMOVED: Slimming to the Death: Herbalife®-Associated Fatal Acute Liver Failure-Heavy Metals, Toxic Compounds, Bacterial Contaminants and Psychotropic Agents in Products Sold in India – J Clin Exp Hepatol. Mar-Apr 2019;9(2):268-272. doi: 10.1016/j.jceh.2018.08.002.
The paper — now removed by the journal — describes a case report of a young woman in India with a BMI of 32.1 (obese) who consumed Herbalife slimming products. According to the now-removed paper, India is the fastest growing market for Herbalife products.
The woman bought the products at a nutritional club that was later shut down by the Government of Kerala. After 2 months of using the products, she developed jaundice, and was admitted to the hospital where she was diagnosed with liver necrosis.
Unfortunately the young woman died while on the waiting list for a liver transplant.
Herbalife analysis in the now-removed paper
Since the authors were unable to obtain the remainder of the slimming products from the family for a toxicology analysis, they bought similar Herbalife products from the same seller in Kerala, India, and from online stores. In total, eight products were subjected to gas chromatography, mass spectroscopy, and other methods for chemical and microbial analysis.
The chemical analysis detected heavy metals, including cadmium, mercury, and lead, as well as other toxic chemicals in all eight products. In addition, 16S rRNA gene sequencing revealed a range of bacterial contaminants in most of the products.
The authors describe similar case reports of other Herbalife-using subjects with hepatotoxicity, and raise the possibility that the heavy metals and bacterial contamination of these unregulated Herbalife products — maybe in combination with unknown toxic phytochemical constituents — could have contributed to these morbidities.
Criticism and legal threats by the company
As per a Twitter thread [archived] by first author Cyriac Abby Philips (@drabbyphilips), several professors working with/at Herbalife or associated companies then requested that the journal should retract the paper because of a lack of evidence.
The authors were asked by the editors to write a scientific rebuttal to the concerns. Both the Herbalife concerns and the rebuttal were published in the journal — but subsequently removed [PDF copy].
Next, a law firm threatened legal action, upon which the journal decided to retract the paper with no clear scientific reason.
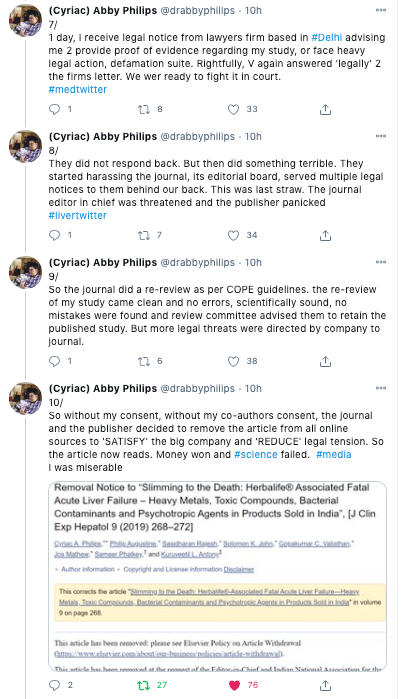
Complete removal of the paper
In a breach of COPE guidelines, and despite the fact that the article had been assigned an issue number ( Clin Exp Hepatol. Mar-Apr 2019;9(2):268-272), the original paper was completely removed by the journal.
COPE guidelines state that the original paper should remain available, but be clearly marked as retracted. Instead, publisher Elsevier removed the paper, and replaced it with a short retraction notice:
“This article, which was published in the March-April 2019 issue of the Journal of Clinical and Experimental Hepatology (“JCEH”), has been removed at the request of the JCEH’s Editor-in-Chief and the Indian National Association for the Study of the Liver (INASL). INASL and JCEH no longer support the content of and conclusions drawn in the article because the scientific methodology, analysis and interpretation of data underlying the article were insufficient for the conclusions drawn, and, with its removal, the article can no longer be relied upon.“
Money wins, science loses
This case is particularly ironic because I have raised scientific concerns about dozens of other Elsevier papers with severe image problems or flawed non-randomized hydroxychloroquine results. None of these concerns have been addressed, and the papers have not been touched.
But as soon as powerful companies threaten to sue a publisher because they are unhappy about a paper, suddenly things get retracted — with no scientific reason.
This is just wrong. Science should be about finding the truth.
I hope the authors can find a publishing house with more courage that will republish their paper.
Update December 22, 2020: Herbalife responds
From an email that Herbalife’s Julian Cacchioli, Vice President, Corporate Affairs, EMEA & India sent me today:
“We were made aware of the above post yesterday – incorrectly suggesting that the decision by Elsevier to remove the original JCEH article was made for non-scientific reasons – which we believe is neither justified nor accurate. It is disappointing that you did not contact Herbalife Nutrition for any comment on this.
The claims made in Dr. Philips’ et al original article were false and unsubstantiated. According to Elsevier’s Policy on Article Withdrawal, ‘in an extremely limited number of cases, … an article [may be removed] from the online database where the article is clearly defamatory, or infringes others’ legal rights, or where the article is, or we have good reason to expect it will be, the subject of a court order, or where the article, if acted upon, might pose a serious health risk.’ The original article contained numerous deficiencies, inappropriate analytical methodologies and incomplete investigative protocols. We presented to the publisher a comprehensive analysis of product testing by three independent international laboratories that undermined the findings of the article. Based on those deficiencies the publisher, Elsevier, took the extraordinary step of removing the article at the request of the Journal of Clinical and Experimental Hepatology (“JCEH”) and the Indian National Association for the Study of the Liver (INASL), stating they ‘no longer support the content of and conclusions drawn in the article because the scientific methodology, analysis and interpretation of data underlying the article were insufficient for the conclusions drawn.’
I am sure you will appreciate the importance of presenting a full and accurate picture, so would ask that you immediately update your site to reflect the above facts. If you have any questions, feel free to contact me.“
Note Elisabeth Bik: The wording provided by Herbalife actually DOES suggest the paper got retracted for non-scientific reasons, so I am not sure how my writing was incorrect.
Update Dec 23, 2020: As pointed out by JLS in the comments, the PubMedCentral version of the retraction notice *does* say the paper got retracted for legal reasons. Here is a screenshot [archived].


I know those some symptoms I’ve been having since I took the product nice enough and got the Frolic in June from Amazon I know is I’ve been having a lot of stomach problems sounds like guess I’ll tell my doctor to do more tests on me they want me to see a specialist on the 28th
LikeLike
All due respect but i also have used for more than a year i have no such issues and i guess the club owners nd representers always tell not to buy any product of herbalife from amzon or flipkart etccc…
LikeLike
Me and my family use Herbalife products for more then 13 years. Both parents feel , look and function (they both work full time job) great, all analysis are normal, both 60+ age. I feel more energetic then before, better eating behaviours, more emotionally balanced ( was addicted to caffeine and sugar before), look younger then my age, my parents and clients also. No disease, less viral disease, better immunity. Also other my friends and clients who consume Herbalife products on a daily basis. My presupposition is that in India there might be fake products market, as it was in Russia 20 years ago. That’ s why many Russians still don’t trust a word Herbalife.
LikeLike
Haha shut up fako
LikeLike
So in layman’s terms, Herbalife said prove your findings or we’ll sue you. They couldn’t so they retracted the paper?
All sounds dodgy to me from the scientists who wrote the paper.
LikeLike
The scientists DID offer proof. Read the post.
LikeLike
I believe these companies are suing because they want proof. There is a scientific process to prove the flaws which lead to the death or deaths. If the editor does not have the scientific proof then there is no there there. Any large reputable newspaper or journal will take on these industries if it turns out the products are killing folks, I mean really that’s how it works. I would want to know as would the public at large and in the US.
LikeLike
Death of the women due to numbers,of complications,covid 19; product is safe,if follow proper guidance.
LikeLike
wow, than I should be dead too. I been consuming those products for over 10 years and my family.
LikeLike
Simply wow!! Such a Nasty company sending people to write fake reviews on a blog post reporting the legal threat to article about their supplements killing people. Surely this company will claim all these paid commenters were just scientists using and praising supplments. This company looks more like a plague in a bottle
LikeLike
This things should be strictly stopped
LikeLike
Been using the product since I was 23yrs, and now I’m 40yrs, my opinion is this is just good food formulated in powders, liquids and tablets form. Same tome
My son been using since he’s in my tummy and he’s 5 yrs old now.
There a lot of misleading in this article, same time herbalife nutrition Manufacture is all in house, to make sure the quality of the product is safe for human to use, a board of scientific advisors besides to create new products same time we have a group of scientists, doctors and nutritionists are there to make sure is matches the world standard and quality of nutrition.
Here’s my suggestion
highly suggest avoid buying Herbalife nutrition product on any E-commerce platforms especially those website selling crazy discount on the products ….. there very high chance those product are fake or expired.
Find a Reliable and trustworthy Coach around your neighbourhood.
Perhaps order the products from herbalife directly
LikeLike
This is exactly what happens when uneducated ******
Note EB: Rest of comment removed because of the presence of some unkind words.
LikeLike
“Feel free to contact me.”
Perhaps you should ask him to confirm whether or not Herbalife have sent any legal threats to any publishers regarding this article? And if so, to send you the text of those letters.
LikeLike
Herbalife themselves said that they sent a legal threat, in the second sentence of the second paragraph of their letter. That sentence also shows that the article was not retracted for scientific reasons. There is no need to ask them for clarification because they make it perfectly clear, but if you want confirmation, look at the current PubMed listing for this article, which now reads: “For legal reasons, the publisher has withdrawn this article from public view.”
LikeLike
Due to the large amount of spam messages praising the company’s product, I will no longer approve those.
LikeLiked by 1 person
Actually, I know Yann (One of the commenters). She’s a normal mom from Hong Kong now living in Malaysia. Also, The fact is Herbalife International sells almost 100,000,000 cans of shake a year, about 250,000 a day, and is approved by 96 different government bodies (Ministry of health in Japan, China, and many other countries). If this product was bad, China of all countries, would never allow an American company sell a bad food product to their people and take money out of the Chinese economy. Remember, China swiftly executed the melamine perpetrators. All these 96 countries strictly test all the Herbalife products for importation. Furthermore, many of the Herbalife ingredients are the same as competing product’s ingredients. So I disagree with your conjecture that people praising the company’s product is spam; it is not. Herbalife is Number 1 worldwide a for a reason. Because it is the best. You do not control 22% of the world weight management market by luck; it’s by a good quality product with good customer service. And there is no greater proof than Herbalife selling 250,000 cans of the Formula 1 shake a day (and if you read the stock report, we grew 12% last year, during COVID…… Nobody is holding a gun to people’s head and told to buy. People are voluntarily handing over their cash because they know it’s worth it, or want to try it. Me, I’ve been eating Herbalife 26 years, liver perfect. Wife just had a liver scan run , perfect. My grandkids also love the shake.
LikeLike
Hello Mike, I was reading your comment and is pretty much what I think about Herbalife. Wish we could talk more about it. I’m about to do a presentation about why say yes to Herbalife and I would love to make some questions if you agree as well.
LikeLike
I thought is was already clear for years that herbalife is a pyramid scheme. When John Oliver did his great segment (https://www.youtube.com/watch?v=s6MwGeOm8iI) in 2016 it felt as news from the earlier century. Is it a surprise that a company that cons other people, is also ready to use mob practices to hide their failures?
* To all the people claiming they have used the herbalife rubbish and did not get a disease: that is the exact false claim used for the ‘healthy’ habbit of smoking. We all know someone that got 80 years old while smoking…
LikeLiked by 1 person
I think it’s clear that most commenters didn’t read or understand the article or they are shills for Herbalife.
It is crystal clear that Herbalife used their shyster lawyers to bully craven Elsevier – not the most upstanding of publishers anyway – into taking the coward’s way out. Shame on you!
So the lawyers found some dodgy labs to say the products were “clean.” Still doesn’t invalidate the tests in the original paper. There are reports in journals going back years on hepatoxicity caused by Herbalife products in countries around the world so this is nothing new. It’s an open secret. The very first paragraph in their Wikipedia entry states this very fact with ELEVEN references!
What is wrong is trying to use legal methods to bully scientific papers into retraction and then claim it’s on the “science.” Just hope Dr Phillips does sue Elsevier and the bad publicity causes the Streisand effect for Herbalife and their MML schemes and their dubious products of very questionable efficacy.
I for one can never understand why anybody buys them – don’t they read the labels?
LikeLike
I m also used this product I realised some side effects on my body.i request government to bann this company.
LikeLike
Many comments on this post seem to come from Herbalife associats. Scientific probe should be conducted on these products.
LikeLike
It’s clear many antagonists of Herbalife company are not users of the products themselves but are merely envious of the successs the company has chalked over the years. We the users know the benefits of the products that is why our slogan is USE THE PRODUCTS, WEAR THE BRAND AND TALK TO OTHERS. NO one consumes a poison and is healthy enough to share his or story with others. Enemies of Herbalife nutrition no one is compelling you to patronise our products so keep your envy and jealousy to yourselves.
Our testimonies have brought the company so far for the past 43 years and it will go on
LikeLike
Thankk you for writing this
LikeLike
coming in late but why would someone be randomly jealous of a global corporate? I follow Dr Cyriac Abby Phillips and had read about this Herbalife issue earlier. A friend told me casually that his wife has started taking some Herbalife stuff. Told him ask her to stop. And today I have shared your blog post Elisabeth Bik. Thank you and thank you to the Liver Doctor for doing all this so people stay safe.
LikeLiked by 1 person